
Happy Friday! Here’s what’s ahead:
Story: Type a Prompt, Get a Drug Candidate
Trial: iBio is betting a single dose can compete in the long-acting obesity race
Research: AI meets organ chips for brain drugs
The Bigger Story
📢 The Lab Just Got a New Employee
Two launches this week want you to know that AI doesn't need your help anymore. Latent Labs debuted Latent-Y, an agent that designs therapeutic antibodies from a text prompt, claiming it compresses weeks of expert work into hours.
Insilico Medicine rolled out PandaClaw, which lets biologists run complex multi-omics analyses by just asking questions in plain English. Both promise autonomous, end-to-end discovery. You type, they science.
Meanwhile, a Sakana AI paper just landed in Nature showing their "AI Scientist" system produced a research paper that passed peer review at a top ML conference workshop. One out of three submissions made the cut. At a workshop with a 70% acceptance rate. So… promising, but let's not print the Nobel citations yet.
The bottleneck in drug discovery was not necessarily that "scientists worked too slowly." It was that most ideas fail, and you don't know which ones until you've burned years and millions finding out. If these agents can make failure cheaper and faster, that changes the math.
We’re basically moving from an "AI-assisted" to "AI-conducted" world. If one researcher can now run dozens of campaigns in parallel, does that change what kind of science gets done, or just how fast? I think we're about to find out how much of that was uniquely human.
Public AI Drug Discovery Companies

BioAge reported a technically "positive" interim Phase 1 data for their lead drug, BGE-102, showing an 86% reduction in heart-risk biomarkers. However, the market seemed to focus on the $80.6M annual net loss from their 2025 Earnings.
For iBio, despite the leadership buying shares (usually a strong bullish signal), the stock is still suffering from a "post-update hangover" following their March 17 cardiopulmonary program update.
BullFrog AI had a new product launch, where they unveiled bfARENAS, AI engine for clinical trial strategy. Another good piece of news, but clearly not reflected in the market.
All others, no real news that stood out in the past week for such major moves.
Brain Booster
At approximately what age does the human brain begin to shrink in volume, potentially leading to cognitive changes?
Select the right answer! (See explanation below and source)
Clinical Trial Snapshot
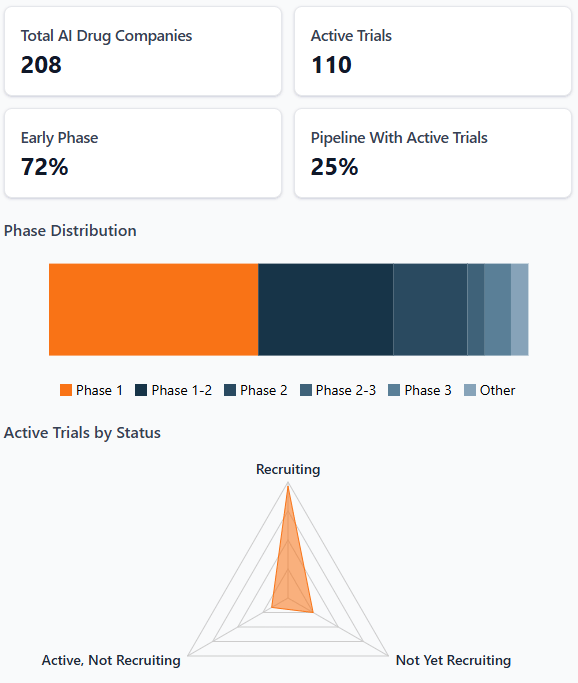
📝 Clinical Trial Updates
AbCellera has moved ABCL575 into a first-in-human Phase 1 trial, dosing healthy volunteers in a single-ascending dose study. The trial is randomized and placebo-controlled across five planned cohorts. AbCellera's AI-driven antibody discovery platform has been behind multiple partnered programs, but details on ABCL575's target and therapeutic area haven't been disclosed. [Link]
iBio has started a first-in-human Phase 1 trial for IBIO-600, its AI-designed obesity candidate, with a nine-month monitoring window after a single dose. The single-ascending dose study in overweight and obese adults will track body composition and physical function changes alongside standard safety and PK readouts. iBio used its AI-driven drug design platform to develop the asset, which appears aimed at the long-acting obesity space. [Link]
What Caught My Eye
A bipartisan bill wants NIST to set national standards for "AI-ready" biological data to accelerate drug discovery. The AI-Ready Bio-Data Standards Act of 2026 would require certain federally funded research to meet AI-readiness criteria and push cross-agency coordination on data formatting and interoperability. The legislation frames biological data as a strategic national asset, citing concerns that fragmented U.S. datasets are falling behind China's centralized AI-biotech data infrastructure. [Link]
The top 16 pharma companies cut R&D spending by 3.6% in 2025, dropping from $165 billion to $159 billion as pipeline prioritization took hold. AbbVie, J&J, and BMS led the pullback through impairment charges and program cuts, while Amgen (+22%), GSK (+18%), and Regeneron (+14%) moved in the opposite direction with increased investment in obesity, oncology, and ADC programs. [Link]
Insilico Medicine is benchmarking frontier AI models head-to-head on their ability to identify clinical-stage cardiovascular drug targets. Their ScienceAIBench series tested GPT-5, Opus 4.1, and DeepSeek R1 across heart failure, ischemia, and pulmonary arterial hypertension, finding that no single model dominated and performance in heart failure was tightly clustered across all models. [Link]
Featured Research
98% of Drugs Can't Get Into Your Brain. Three Technologies Are Trying to Fix That.

Neurological diseases now affect 3.4 billion people globally, making them the leading cause of disability worldwide. You'd think that would attract massive pharmaceutical investment, well, it hasn't.
The brain's blood-brain barrier blocks more than 98% of small-molecule drugs from entering the organ they're supposed to treat, and the Phase II and III failure rate for CNS drugs sits at approximately 85%, second only to oncology. Big pharma has responded the way you'd expect… by walking away. Over the past decade, several major companies have effectively shuttered their neuroscience divisions.
A new review in Frontiers in Pharmacology argues that the field's rescue plan is already being assembled. Chen et al. map a convergence of three technologies that, until recently, existed in separate silos: AI, microfluidics, and nanodelivery.
Separately, each is useful. Together, they start to look like a closed-loop system that could fundamentally change how we discover and deliver drugs for the brain.
Here's the quick version of how they fit. Microfluidic "brain-on-a-chip" platforms reconstruct the blood-brain barrier, neuron-glia interactions, and synaptic connections on a device the size of a USB drive. They let researchers test whether a drug candidate actually enters the brain before committing to animal studies or clinical trials.
The FDA announced in April 2025 that it intends to replace animal-testing requirements with new approach methodologies, including organ-on-chip technology, within three to five years. Companies like Emulate and AxoSim are already building these platforms for neurology specifically.
Then there's AI-driven nanocarrier design. Instead of manually optimizing drug-carrying nanoparticles for size, surface charge, and targeting ligands through trial and error, teams are using graph neural networks and generative models to predict how particles will interact with barrier endothelial cells and release their payloads.
The review highlights reinforcement learning algorithms that can simulate nanoparticle trajectories through vascular networks and reverse-engineer the optimal morphology for reaching a brain lesion. The AI literally optimizes the shape and stiffness of a nanoparticle to maximize enrichment at the target site.
The most ambitious part of the paper describes what happens when you close the loop: AI reads real-time imaging data from nanocarriers in vivo, detects when they're not reaching the target, and triggers focused ultrasound to transiently open the barrier in exactly the right spot.
The review calls this "cruise therapy," and while that label is aspirational, Bristol Myers Squibb has already partnered with Insitro to use AI for identifying previously unknown biology in ALS, and NVIDIA and Eli Lilly launched a $1 billion co-innovation lab in January 2026 to build exactly these kinds of closed-loop, lab-in-the-loop AI systems for drug discovery.
While the idea is nice, most of this work remains preclinical. Only a few nanoparticle formulations have entered clinical trials for brain-targeted delivery, and the clinical translation of nanomedicine-based therapies for neurological diseases remains in its early stages.
The AI models that design these systems are still black boxes, and biologists don't trust design rules they can't trace to known biochemistry. The gap between a brain chip in a lab and a treatment in a patient's skull is enormous.
But there could be some hope! The FDA's April 2025 roadmap stated that animal use should become "the exception rather than the rule," and the agency may offer streamlined reviews for companies using alternative methods.
Organ-chip companies are scaling up accordingly. The question this review leaves hanging is whether these three technologies will actually integrate in practice, or stay in their silos, publishing parallel papers that cite each other but never share a lab bench.
Sources: [Research Article]
Have a Great Weekend!

❤️ Help us create something you'll love—tell us what matters!
💬 We read all of your replies, comments, and questions.
👉 See you all next week! - Bauris
Trivia Answer: B) 30 years old
The human brain reaches its maximum volume in early adulthood, but subtle atrophy (shrinkage) begins as early as the third decade of life (around age 30). This process is gradual and affects regions like the frontal lobe and hippocampus, which are involved in memory, planning, and decision-making. While these structural changes start early, noticeable cognitive decline typically occurs much later, as the brain compensates through plasticity and redundancy. [Source]


