
Happy Friday! Here’s what’s ahead:
Story: The AlphaFold era of sharing may be over
Trial: China advances an AI-designed obesity drug to Phase III
Research: Stanford built a virtual biotech for 46 dollars
Once in a while, the right words make the problem impossible to ignore. Deepmirror founder on Meet The Drapers (a VC reality show, yes it’s a thing): 20,000 cancers, ~1,000 drugs. That's it. That's the whole pitch for AI in drug discovery.
The Bigger Story
📢 The Open-Source Era of AI Drug Discovery May Be Ending
Isomorphic Labs earlier this month published a 27-page technical report about their new drug-discovery AI, IsoDDE, and the scientific community's reaction was basically: "This looks incredible. Now can you tell us literally anything about how it works?" They cannot, or rather, they won't.
That's a meaningful departure from their original spirit of science approach. AlphaFold 2 won a Nobel Prize partly because DeepMind gave it away. IsoDDE, which researchers are calling "an AlphaFold 4," is staying locked behind the Isomorphic door.
The problem here is that Isomorphic has billion-dollar deals with Lilly, Novartis, and J&J.
They're building their own clinical pipeline. They're reportedly licensing proprietary structural data. The incentive to share has obviously changed. Why publish your secret sauce when the sauce is the business?
Open-source teams like MIT's Boltz project are pushing back, and making real gains with public data. So the race is on.
But for the future, you do have to ask yourself, if the most powerful drug-discovery models become proprietary infrastructure owned by a handful of well-capitalized labs, what happens to the academic science that made them possible in the first place?
For more details: Full Article
Public AI Drug Discovery Companies
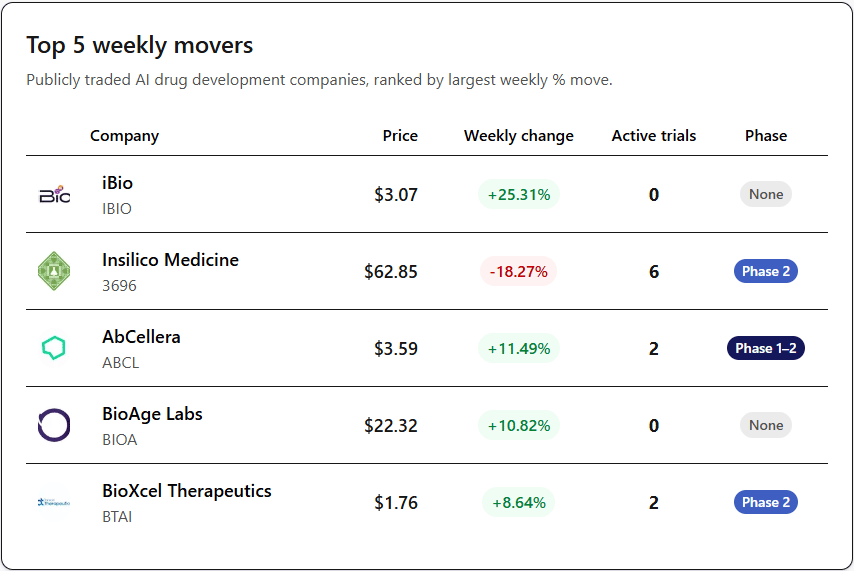
Insilico’s drop is a weird one because the headlines have been mostly positive, so it reads like profit-taking and general Hong Kong biotech volatility rather than a specific bad-news event.
Brain Booster
Which gene is most commonly mutated in human cancers and is often called the “guardian of the genome”?
Select the right answer! (See explanation below and source)
Clinical Trial Snapshot
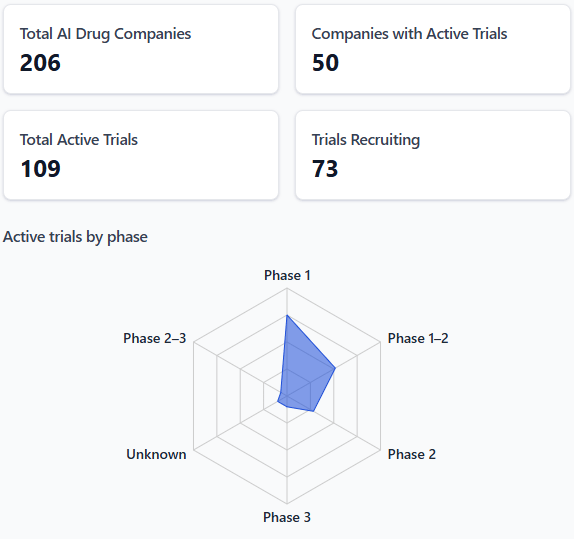
📝 Clinical Trial Updates
MindRank dosed its first patient in a Phase III trial for an AI-designed GLP-1 drug, making it one of the furthest-along fully AI-generated molecules in the clinic. The company's Molecule Pro platform designed MDR-001, a biased GLP-1 receptor agonist, reaching IND filing in 19 months. The MOBILE trial will enroll roughly 750 participants with obesity and type 2 diabetes across 50 sites. [Link]
Juvenescence completed a Phase 1 trial for its AI-enabled PAI-1 inhibitor, a first-in-class drug targeting fibrotic and metabolic diseases linked to aging. The small molecule showed a good safety and tolerability profile with once-daily dosing, and the program is backed by human genetic data linking reduced PAI-1 to a 10-year lifespan extension. A Phase 2 proof-of-concept trial is planned for later in 2026. [Link]
What Caught My Eye
Insilico's CEO just called Recursion the only real AI drug discovery company on the public markets. Alex Zhavoronkov posted on LinkedIn that Recursion is the "only real publicly-traded alternative" in AI drug discovery, dismissing others as "mostly pure biotechs." He argued that AI drug discovery companies should ultimately be judged as more efficient biotechs that need to produce therapeutics faster, cheaper, and at higher quality. [Link]
The FDA's 2026 guidance agenda includes new rules for using AI and machine learning in pharmaceutical manufacturing. CDER's agenda lists 81 guidelines scheduled for release this year, with a new addition specifically covering AI/ML quality considerations in pharma manufacturing. The agenda also adds guidance on digital health technologies in clinical investigations and distributed manufacturing. [Link]
An autonomous lab system built by OpenAI and Ginkgo Bioworks outperformed a PhD student's protein synthesis optimization by 40%. The system used GPT-5 to interpret results and design experiments executed by Ginkgo's lab robotics, testing over 30,000 conditions across six months compared to the student's 1,231 combinations. The Nature article notes that current lab robotics still can't handle tasks requiring dexterity or bespoke experiments like tissue or animal work. [Link]
Featured Research
Stanford Built a Virtual Biotech Company, and It Costs $46 to Run
A team at Stanford just published a preprint called "The Virtual Biotech." They built a multi-agent AI system that mirrors the structure of an actual drug discovery organization, complete with a virtual Chief Scientific Officer, specialized scientist agents for genetics, single-cell biology, clinical trials, and safety, and over 100 analytical tools.
Think of it as an AI org chart that can interrogate real biomedical databases, run analyses, argue with itself, and produce a full target assessment report. For $46 in API costs.
To show the system’s capabilities, they pointed these agents at B7-H3, a protein in lung cancer, and asked it to evaluate whether it's a viable drug target.
The AI independently analyzed genetics, single-cell data, spatial transcriptomics, and clinical survival data, then concluded that B7-H3 was most promising as an antibody-drug conjugate (that's a targeted therapy that delivers a toxic payload directly to cancer cells).
It even identified that B7-H3 was overexpressed not just in cancer cells but in the surrounding fibroblasts, something the existing literature hadn't really emphasized.
The really interesting part is that the AI's model had a knowledge cutoff of January 2025. Months later, Merck and Daiichi Sankyo reported that ifinatamab deruxtecan, a first-in-class B7-H3 ADC, achieved a 48.2% response rate in a Phase II trial for small cell lung cancer, and the FDA granted it Breakthrough Therapy Designation in August 2025.
The AI couldn't have known that. It arrived at the same strategy from the data alone.
They also had it analyzed 56,000 clinical trials and found that drugs targeting cell-type-specific genes were 48% more likely to reach market and had 32% lower adverse event rates. That's the kind of insight that usually takes years of meta-analysis by large research teams.

Stanford's Virtual Biotech analyzed 56,000 clinical trials autonomously, each one processed by its own AI agent. The attrition you see here is exactly the problem the system was built to solve.
Now, the caveats. This is a preprint, not peer-reviewed. I’m sure the AI agents can still make errors that propagate through the system. And "arriving at the same conclusion as pharma companies" is not the same as discovering something nobody knew. The real test will be whether systems like this can find targets that humans miss entirely.
But I have to come back to the cost. Forty-six dollars. A comparable human-led assessment would involve a cross-functional team working for weeks or months.
If this kind of analysis becomes routine, the bottleneck in drug discovery won't be intelligence or data. It'll be whether anyone trusts the machine enough to act on what it finds.
Sources: [Research Article]
Have a Great Weekend!

❤️ Help us create something you'll love—tell us what matters!
💬 We read all of your replies, comments, and questions.
👉 See you all next week! - Bauris
Trivia Answer: D) TP53
TP53 is the most frequently mutated gene in human malignancies, 50% of which carry alterations to it. TP53 helps stop damaged cells from dividing (or triggers cell death), so when it’s mutated, cells can grow unchecked. [Source]

