
Happy Friday! Here’s what’s ahead:
Story: Google's Secretive Takeover of the FDA
Trial: Lantern’s AI-designed LP-184 closed Phase 1a enrollment at 64 patients
Research: Big AI Lost the Drug Discovery Benchmark
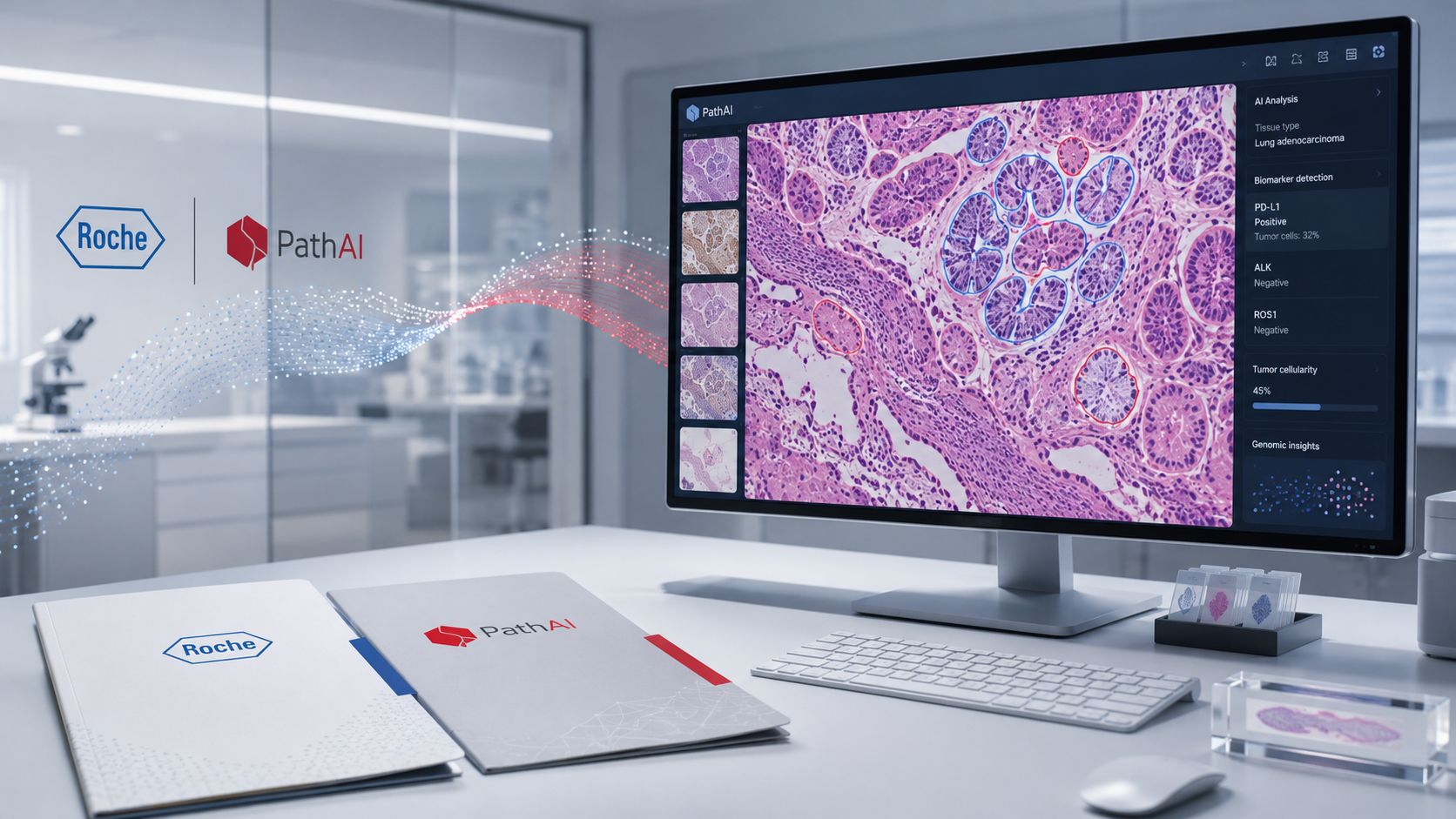
Roche and PathAI started partnering in 2021, scaled it up in 2024, and this week made it official with a $750M acquisition plus up to $300M in milestones. Five years of dating, then the ring. Pharma's love language is companion diagnostics!
The Bigger Story
📢 Google's Secretive Takeover of the FDA
The FDA just announced Elsa 4.0 alongside HALO, a new system consolidating more than 40 of the agency's data sources into one place. Commissioner Makary said the upgrades will make staff workflows "more efficient and enjoyable," which means they're rebuilding how the agency runs from the inside out.
The line that actually matters came from the Chief AI Officer: "Elsa sits on top of our data."
Until now, Elsa was a chatbot that reviewers fed documents to. Now, it's the interface between FDA staff and the agency's own systems is becoming an AI.
It's also running on Google Cloud. Which, if you've been tracking this, follows the FDA's migration off Claude earlier this year. Google now owns the regulator's stack from the data layer up.
So, if the tool sits on top of the data, the reviewer's job will change from reading files to trusting summaries. Is that still review?
For more details: Full Article
Public AI Drug Discovery Companies
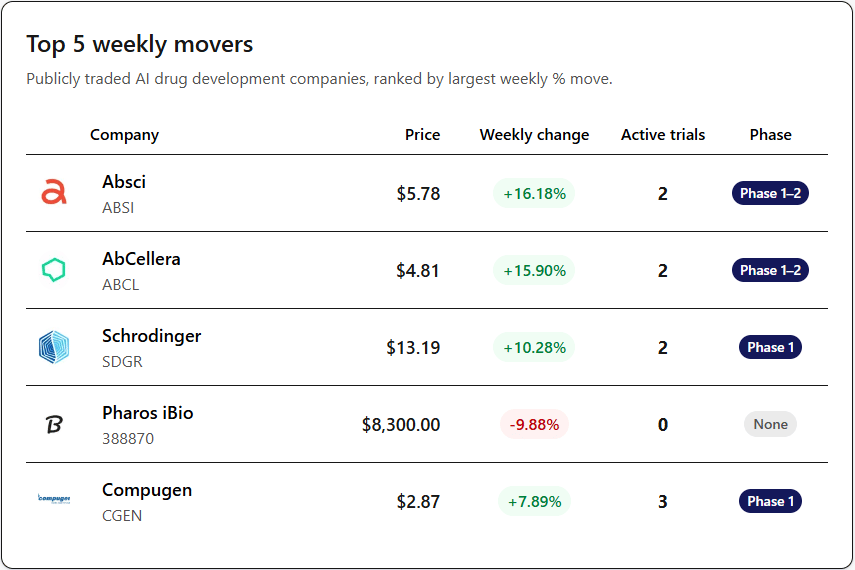
On April 20, AbCellera confirmed it would release interim Phase 1 results for its "first-in-class" menopause drug this coming Monday. So, investors are probably piling in this week to catch the move before they reveal that highly anticipated Phase 1 data.
Schrodinger crushed their revenue targets this week, mostly because their drug discovery business is growing like crazy. Revenue reached $58.6M (21% above estimates), which more than doubled YoY.
Brain Booster
Which street dance style is known for its angular, geometric arm and hand movements that resemble poses from ancient Egyptian art?
Select the right answer! (See explanation below and source)
Clinical Trial Snapshot

📝 Clinical Trial Updates
Lantern Pharma's LP-184 has officially closed enrollment at 64 patients, clearing the way for biomarker-guided Phase 1b/2 trials in TNBC, KEAP1/STK11-mutant NSCLC, and PTGR1-overexpressing bladder cancer. The Phase 1a, designed using Lantern's RADR AI platform, hit all primary endpoints with a 48% clinical benefit rate at or above the therapeutic dose threshold. LP-184 holds FDA Fast Track designations for GBM and TNBC, plus four Rare Pediatric Disease designations. [Link]
What Caught My Eye
An AI tool deployed by the Institute of Physics Publishing just flagged 2,500 suspicious peer-review reports out of roughly 500,000 submitted between 2020 and 2025. The Duplicate Review Checker is the first AI system specifically designed to catch plagiarized or template-generated peer reviews, identifying 785 reports with at least 80% overlap and 89 exact duplicates. PLOS separately investigated about 150 papers for peer-review integrity issues between 2024 and 2026 and retracted 40, with its head of publication ethics calling it "the tip of a very large iceberg." [Link]
A UK consortium led by Diamond Light Source just released OpenBind v1, an open-access AI model and dataset aimed at becoming the "AlphaFold for protein-drug binding." The first release contains 800 high-resolution binding measurements generated in seven months through Diamond's XChem fragment screening facility and the UK's Isambard-AI computing cluster. The project, funded by the UK Department for Science, Innovation and Technology, is meant to fill the experimental data gap that has held AI drug discovery back the way PDB enabled AlphaFold2. [Link]
The New England Journal of Medicine just issued its first retraction since 2020 after authors admitted using AI to alter a clinical image. The April 18 "Images in Clinical Medicine" piece showed black airway casts removed from an 87-year-old man with forest-fire lung damage, with a measuring tape the authors acknowledged repositioning using an AI tool. A PubPeer commenter flagged the manipulation by noting the ruler's numbers did not count correctly, and the journal pulled the article on April 29. [Link]
Featured Research
The 25-Year-Old Algorithm Beating Drug Discovery's Foundation Models

Over the past four years, the AI-for-drug-discovery world has produced more than 200 "molecular foundation models," giant networks pretrained on millions of compounds with the implicit promise that scaling beats specialization.
A new bioRxiv preprint from the Global Health Drug Discovery Institute tested that promise across 22 drug discovery prediction tasks, ran 167,000 separate evaluations through them, and produced a result the field has been suspecting. The foundation models lose more often than they win.
Classical machine learning models, the ones that fit a few hundred decision trees over old-school chemical fingerprints, won 10 of the 22 tasks. Graph neural networks, which treat each molecule as a network of atoms and bonds, won 9. The big pretrained "chemical language" models, the multi-million-parameter networks supposed to transfer knowledge from massive corpora of compounds, won only 3.
On the tuberculosis prediction task specifically, a random forest fed with classic molecular fingerprints beat the foundation model outright.

Across 22 drug discovery prediction tasks, the field's smallest model class wins as many tasks as every other family combined. Pretrained molecular foundation models, the centerpiece of the AI drug discovery investment thesis, take a minority slice in every category.
The benchmark also tested large language models prompted to extract medicinal chemistry rules from training molecules and then apply those rules to held-out compounds. They won zero tasks.
The rules themselves looked chemically sensible, recovering known motifs like nitro groups for mutagenicity and quinolines for antimalarial activity, but as standalone predictors they couldn't beat simple statistical models trained on the same data.
The timing is awkward for AI drug discovery's marquee names. Recursion shelved three advanced programs in May 2025, including the cerebral cavernous malformation candidate that had been its flagship AI-designed drug.
BenevolentAI's eczema drug failed Phase IIa in 2024.
Insilico's lung fibrosis program is the bright spot, heading for Phase III after positive mid-stage data. The story that bigger pretrained models can reliably translate into better drugs is harder to sustain in 2026 than it was in 2022, and benchmark papers like this one are part of why.
Just something to keep in mind, however, this is a single-author preprint, not yet peer reviewed, from a TB-and-malaria-focused institute that built two of the four datasets it evaluates on.
Other 2025 benchmarks like Polaris have shown newer descriptor-based foundation models such as CheMeleon outperforming random forests on different task suites, so the field's verdict is far from settled. And the paper's "GPT5.5-SAR" and "Opus4.7-SAR" labels are misleading: those are rule-extraction workflows attributed to those models, not live frontier reasoning.
The honest reading is "rules extracted from LLMs don't beat trained predictors," not "LLMs can't do chemistry."
Most of the value of pretraining a 47-million-parameter chemical language model seems to evaporate when the held-out test set is genuinely structurally different from training molecules, which is the only kind of test that matters for real drug discovery.
The path forward may not be more pretraining data. It may be better molecular representations, more honest benchmarks, and that some 25-year-old algorithms are still good enough.
Sources: [Research Article]
Have a Great Weekend!

❤️ Help us create something you'll love—tell us what matters!
💬 We read all of your replies, comments, and questions.
👉 See you all next week! - Bauris
Trivia Answer: C) Tutting
It’s known for angular, geometric movements of the arms, hands, and fingers, often resembling the stiff, squared-off poses seen in ancient Egyptian art.

