
Happy Friday! Here’s what’s ahead:
Story: Anthropic Wants to Be the Drug Company
Trial: Recursion revives a shelved LSD1 drug
Research: Custom Cancer Drugs in a Month?
The Bigger Story
📢 Anthropic Wants to Be the Drug Company
If you were anywhere near a timeline this week, you saw it. Anthropic bought a stealth biotech startup called Coefficient Bio for $400 million in stock. A company founded in 2025, six employees, no public product. That's roughly $67 million per headcount, which is corporate-speak for "we're buying the people and the thesis, not the revenue."
Coefficient slots into an expansion Anthropic has been telegraphing for months. Claude for Life Sciences launched in October, Claude for Healthcare in January, and leadership has called life sciences one of the company's largest strategic moves.
OpenAI is running the same play with ChatGPT Health and its healthcare enterprise suite.
The foundation model labs aren't content to sell API access to pharma anymore. They're hiring the biologists, absorbing the startups, and building vertical stacks aimed straight at drug discovery.
All of this lands the same week HHS banned Claude for federal employees, a move that could ripple into the FDA's Elsa review tool built on top of it. Anthropic is going deeper into life sciences precisely as one of its biggest potential government customers slams the door.
So if the labs are becoming drug discovery companies, what's left for the drug discovery companies that were betting on them as partners?
For more details: Full Article
Public AI Drug Discovery Companies
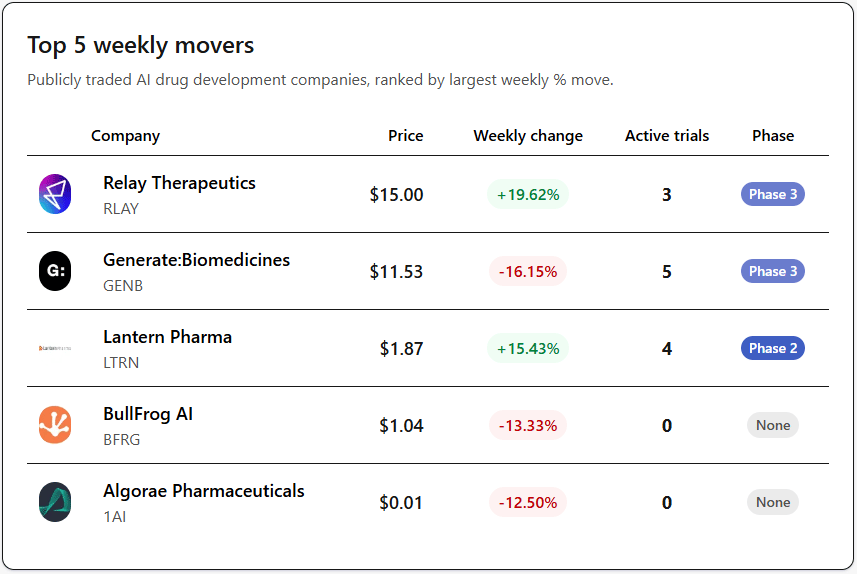
Relay (+19.62%) climbed ahead of an upcoming Zovegalisib readout at a world congress, with analysts circling $17–$22. A CFO stock sale on April 7 barely registered, which tells you the market is pricing the data.
Lantern Pharma (+15.43%) finally shipped. Their April 9 live demo of withZeta.ai, pitched as the first multi-agentic co-scientist for rare cancers, put a working product in front of the market for the first time.
Brain Booster
Which of the following symptoms is more commonly seen in women experiencing a heart attack compared to men?
Select the right answer! (See explanation below and source)
Clinical Trial Snapshot
📝 Clinical Trial Updates
BioXcel just completed a small validation study for its agitation drug BXCL501, one of the few AI-repurposed drugs to reach the market. The 30-patient open-label study (NCT07116694) tested whether patient- and informant-rated agitation scales align with clinician ratings across schizophrenia and bipolar spectrum disorders. [Link]
Recursion has quietly restarted REC-4539, the Exscientia-designed LSD1 inhibitor it paused last year during its post-merger pipeline cleanup. A new Phase 1 dose-escalation study (NCT07517198) in roughly 40 patients with small cell lung cancer, NSCLC, prostate, ovarian, TNBC, and other solid tumors went live this week. [Link]
Athos Therapeutics just posted its Phase 2b trial for ATH-063, an AI-designed G9A inhibitor heading into biologic-refractory ulcerative colitis patients. The 120-patient, 12-week placebo-controlled study (NCT07513181) is scheduled to start in May 2026 and targets UC patients who've failed existing advanced therapies. [Link]
What Caught My Eye
Massive Bio's AI trial matching system just posted prospective results from 3,804 cancer patients, cutting oncologist match time from 120 minutes to 30. The peer-reviewed study generated over 17,000 oncologist-confirmed trial matches during routine clinical practice in 2024, not a retrospective chart review. The underlying system combines large language models with an oncology-specific knowledge graph and clinician oversight, an architecture Massive Bio argues is more auditable than standalone LLM approaches. [Link]
Evaxion just wrapped the one-year extension of its Phase 2 trial for EVX-01, an AI-designed personalized cancer vaccine that posted a 75% objective response rate in advanced melanoma. 12 of 16 patients had clinical responses and 92% were still responding at the two-year mark, with no relapses observed. [Link]
Tempus just expanded its oncology R&D collaboration with Gilead to an enterprise-wide deal, giving Gilead access to the full Lens AI platform and Tempus' multimodal data library. Gilead has already been using Tempus data for trial design, indication selection, biomarker strategy, and real-world evidence work, and the new agreement broadens that access across multiple indications and bundles in dedicated Tempus analytical services. Financial terms of the multi-year deal were not disclosed. [Link]
Featured Research
A Cancer Drug Designed for One Patient, Manufactured in a Month. Here's What's Stopping It.
A new Perspective in ACS Nano Medicine sketches out a tidy little vision: a tumor biopsy on Monday, a custom-designed cancer drug on the IV pole four weeks later. The authors call it the N-of-1 convergence workflow, and the whole point of the piece is to argue that the AI tools needed to pull this off already exist and are just waiting to be wired together.
The pitch rests on three things that have all matured in the last 18 months.
First, there's now an AI model from Google DeepMind that can read a patient's tumor DNA and predict how a single mutation rewires the way nearby genes turn on and off. That matters because it can flag rare, patient-specific proteins that exist only on that one person's cancer cells.
Second, a separate set of AI tools can design a brand-new protein from scratch to grab onto those targets, in days, with the kind of binding strength that used to take years of lab work.
Third, these custom-designed proteins get glued onto an off-the-shelf nanoparticle in an automated chip-based system, then tested against a miniature version of the patient's tumor grown in a dish before anyone gets injected with anything.
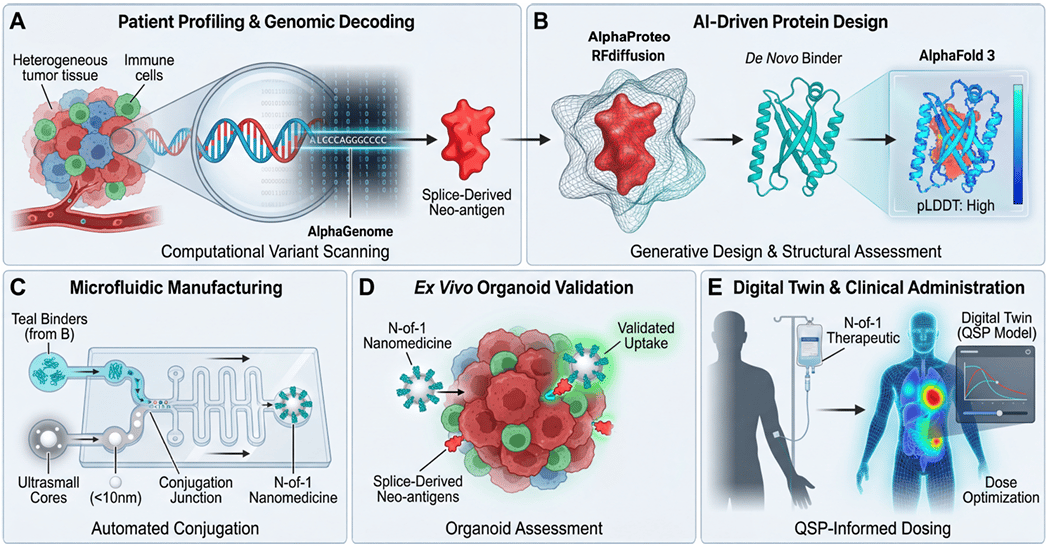
The whole pitch in one diagram. A tumor biopsy on Monday, a custom-built cancer drug on an IV pole four weeks later. Every tool in this loop now exists. Whether they connect in the real world is the open question.
It’s worth noting that senior author Michelle Bradbury co-founded Elucida Oncology, the Weill Cornell spinout that makes the ultrasmall silica nanoparticles she happens to nominate as the off-the-shelf chassis for this whole pipeline.
Elucida's lead drug is currently in Phase 1/2 trials for ovarian, breast, and lung cancer. So this perspective is part roadmap, part business plan. That doesn't make it wrong, but you should read it knowing the author has a horse in the race.
The reason any of this matters is that personalized cancer therapy already sort of exists, and it's painfully slow. Moderna and Merck's custom mRNA cancer vaccine cut melanoma recurrence by 44% in a recent trial. BioNTech's pancreatic cancer vaccine kept eight of sixteen patients cancer-free for over three years. Both work. Both also take six to nine weeks to manufacture per patient and cost more than $100,000 a dose.
The Bradbury pipeline is essentially arguing that swapping a custom protein onto a reusable nanoparticle is faster and cheaper than encoding the whole thing in mRNA every time.
The honest weak spots are exactly the ones you'd expect. AI-designed drugs sail through Phase 1 safety trials at unusually high rates, but their Phase 2 efficacy success is still stuck around 40%, the same as everything else.
Proteins designed from scratch can sometimes trigger the patient's immune system to attack the drug itself, which has shown up in roughly half of similar engineered proteins. And the FDA's manufacturing rules were written to validate one fixed product made the same way every time, not a pipeline that spits out a different molecule for every patient.
That last one is the real wall. The molecules are almost ready, but the paperwork isn't even close. For four-week personalized cancer drugs to actually happen, regulators have to start validating processes instead of products, which is a philosophical shift, not a technical one.
So the question this piece raises is whether the FDA's framework can bend before the patients waiting on it run out of time.
Sources: [Research Article]
Have a Great Weekend!

❤️ Help us create something you'll love—tell us what matters!
💬 We read all of your replies, comments, and questions.
👉 See you all next week! - Bauris
Trivia Answer: B) Jaw pain and unusual fatigue
While chest pain is a common heart attack symptom, 43% of women don’t experience it at all. Instead, they may have jaw pain, nausea, extreme fatigue, or shortness of breath, which are often mistaken for stress or indigestion. This can delay treatment and increase risk. Recognizing these symptoms can save lives! [Source]


