
Happy Friday! Here’s what’s ahead:
Story: 9,000 Jobs Out, One OpenAI Deal In
Trial: BigHat posts its first human cancer trial
Research: AI is now building virtual cell models
The Bigger Story
📢 The Most Expensive Restructuring Narrative in Pharma
Novo Nordisk just announced a "strategic partnership" with OpenAI to "accelerate drug discovery, improve clinical development, and deliver new medicine to patients faster than ever before…"
But read past the press release, and you’ll realize something is not adding up. Novo cut 9,000 jobs to save $1.25 billion annually. CEO Mike Doustdar told Reuters the OpenAI deal would "slow the pace of future hiring." And the partnership's actual focus? Manufacturing, supply chain, distribution, and commercial operations. It’s not exactly the bleeding edge of drug discovery.
What IS important to remember is that Novo already has a serious adoption of AI. Anthropic's Claude powers their clinical study report system, supposedly cutting creation time from 12 weeks to 10 minutes. They're running custom models on Denmark's Gefion supercomputer. Over 25,000 employees use internal AI chatbots.
So, is the OpenAI deal really about building AI capability, or is it about giving the restructuring a forward-looking narrative? Every pharma company needs an AI story right now. But when "AI partnership" looks a lot like "we're automating the roles we just cut," who's the audience for the press release, investors or scientists?
For more details: Full Article
Public AI Drug Discovery Companies

Lantern Pharma, is seeing major gains following the commercial launch of its withZeta.ai platform, which opens up a new subscription revenue stream alongside its clinical pipeline.
AbCellera, is catching a bid after analysts pointed to its $700 million cash fortress and "undervalued" pipeline as a safe harbor for investors.
Brain Booster
What is the real reason why you sometimes wake up completely paralyzed and unable to move, even though you're fully conscious?
Select the right answer! (See explanation below and source)
Clinical Trial Snapshot
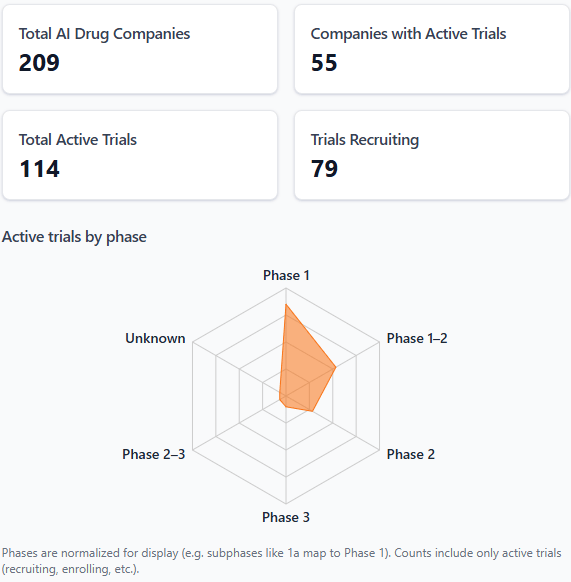
📝 Clinical Trial Updates
BigHat Biosciences just became a clinical-stage company, posting its first-in-human trial for an AI-designed antibody-drug conjugate targeting gastric cancer. BHB810, a CDH17-directed VHH-Fc ADC engineered using BigHat's Milliner AI platform, cleared 13 preclinical gastric tumor models with complete or near-complete responses, per data presented at AACR 2026. The smaller single-domain antibody format was AI-optimized for deep tumor penetration and zero detectable payload leakage in serum stability testing, with first patient dosing expected June 2026. [Link]
iRegene Therapeutics is now recruiting for a Phase I/III trial testing an iPSC-derived cell therapy in multiple system atrophy, a neurodegenerative disease with no approved treatments. NouvNeu001, a single intracerebral injection of lab-grown dopaminergic progenitor cells built using iRegene's AI-plus-chemical-induction platform, will be evaluated for safety, tolerability, and preliminary efficacy in MSA-P patients. [Link]
What Caught My Eye
OpenAI launched GPT-Rosalind, a purpose-built reasoning model for drug discovery and life sciences research, with Amgen, Moderna, and Novo Nordisk among its early access partners. The model leads on BixBench for bioinformatics tasks and ships with a Codex plugin connecting to 50-plus scientific databases and tools for workflows like sequence analysis, literature review, and experimental planning. Access is currently limited to qualified US enterprise customers through a trusted-access program with no token costs during the research preview. [Link]
A new Medicare pilot program using AI to review and deny claims has sparked a congressional pushback, with legislation introduced to shut it down. The WISeR program, active in six states since January, pays private companies a share of costs "saved" by rejecting claims, and 80% of similar denials in Medicare Advantage were overturned on appeal in 2024. Rep. DelBene has introduced a bill to end the program, calling it a vehicle for privatizing traditional Medicare. [Link]
Demis Hassabis said his original motivation for AI was to cure cancer, not fuel a commercial race. In a recent interview with YouTuber Cleo Abram, the Google DeepMind CEO said he entered AI to harness the world's data for fundamental scientific questions, contrasting that vision with the industry's current trajectory. [Link]
Featured Research
An AI Built the AI That Figures Out How Cells Respond to Drugs

About 90% of drugs fail in clinical trials, mostly because the cellular response you get in a lab doesn't match with what happens in a human body. That's the problem "virtual cells" are supposed to solve.
You feed a model a drug or a gene knockout, and it predicts how the cell's gene expression will respond. Get that right, and you can screen millions of ideas virtually before ever walking into a lab.
This has become one of the hottest races in computational biology.
The Arc Institute released its State model in June 2025, trained on 167 million observational cells and over 100 million perturbed cells across 70 cell lines. Their inaugural Virtual Cell Challenge, sponsored by NVIDIA, 10x Genomics, and Ultima, offered a $100,000 grand prize and drew 1,200 teams from 114 countries.
In January 2026, Tahoe Therapeutics, Arc, and the Chan Zuckerberg Biohub announced a multi-million dollar partnership to generate 120 million more single-cell data points specifically for training virtual cells. Xaira Therapeutics, a $1 billion AI drug discovery startup, dropped its own massive Perturb-seq dataset last year to fuel the effort.
All of this effort pours into models that are themselves painful to build. An ML team picks an architecture, tunes it, debugs it, tries fifty variants, and six months later ships something that works on one cell type. The real bottleneck is how many architectural ideas a human team can try.
A new preprint from GenBio AI, a competitor to Arc in the virtual cell space, challenges that. They built VCHarness, a system where a coding agent, Claude Sonnet 4.6 specifically, designs the models itself. It writes the code, trains the model on H100 GPUs, reads the performance numbers, and decides what to try next. The whole loop runs for days instead of months.
Across four different cell types, VCHarness beat every human-designed baseline at predicting which genes would go up or down after a CRISPR knockout. Performance jumped from roughly 0.44 to 0.51 on the standard metric, a meaningful gain in a field where decimals are hard-won. The architectures the AI converged on weren't what humans would have built. The paper calls them "non-obvious."
DeepMind started this trend last year with AlphaEvolve, an AI system that used Gemini to design better algorithms for Google's own infrastructure, including the kernels that train Gemini itself. VCHarness is the biology version of that loop.
Foundation models designing the foundation models that understand cells…
All of this is very promising, but it’s important to note that the benchmarks are CRISPR knockouts in immortalized cell lines, a narrow slice of what "virtual cell" actually needs to mean for drug discovery.
Predicting responses in primary human tissue, across diseases, with real small molecules, over time, none of that is tested. Right now, it’s useful for research, but nowhere near clinically predictive.
Sources: [Research Article]
Have a Great Weekend!

❤️ Help us create something you'll love—tell us what matters!
💬 We read all of your replies, comments, and questions.
👉 See you all next week! - Bauris
Trivia Answer: B) You're briefly trapped between sleep stages.
During REM sleep (when you dream), your brain intentionally paralyzes most of your voluntary muscles to stop you from physically acting out your dreams. Sleep paralysis happens when you wake up mentally but your body hasn't yet exited that REM-induced paralysis. Scary!


